Public trust and confidence in the efficacy, safety, and appropriateness of a coronavirus vaccine to be procured and made available in a country like the Philippines is heavily dependent on the political context in which a potential candidate vaccine is approved and distributed.
These are also contingent on the kind of information about the potential candidate COVID-19 vaccine(s) being dispersed by the mainstream media, medical practitioners, political personalities, and governments.
The more politicised the COVID-19 vaccine procurement and process becomes, combined with widespread fake news, misleading information, and half-truths, the more public health and the COVID-19 situation in a country becomes problematic and highly risky.
In the context of the Philippines, the black propaganda, and politicisation of the COVID-19 vaccine procurement against the Philippine government and the spread of half-truths, fake news, misinformation/disinformation, and misplaced criticisms against Chinese COVID-19 vaccines (i.e., Sinovac and Sinopharm) by mainstream media and by certain political personalities belonging to the political opposition are not just insidious but downright outrageous.
Misleading Information And Half-Truths
For instance, the Philippine Daily Inquirer (PDI) reported that the Department of Science and Technology (DOST) had approved the “50-percent efficacy” of the Sinovac vaccine. This is half-true and misleading. The statement of Senate Majority Leader Juan Miguel Zubiri in a PDI news article saying that “An efficacy rate of 50 percent for a vaccine against coronavirus is “a joke” and “totally unacceptable,” was again not only another misleading but also incorrect statement too.
Zubiri remarked after the DOST had said that the “50 percent efficacy” of the COVID-19 vaccine from China’s Sinovac Biotech is “acceptable” and within the World Health Organization’s (WHO) minimum requirement.
Likewise, statements coming from the likes of Dr Anthony “Tony” Leachon, a former special adviser to the National Task Force (NTF) Against COVID-19, who was removed from his post in June 2020 by President Duterte because of the manner in which he had cast doubts on the Department of Health’s (DOH) COVID-19 reporting data, and is now a disparaged critic of the government, were also misleading.
Leachon is asking the Philippine government why it is allegedly settling for a vaccine with a 50 percent efficacy rate (referring to Sinovac). He said, “If Pfizer and Moderna boast of 95 percent efficacy rate and AstraZeneca has 70 percent, why would the Philippines settle for Sinovac 50 percent efficacy rate and much more expensive than Pfizer and Astra?”
He also added that, “We tolerate mediocrity. If other countries would like to have world-class vaccines with a 95 percent efficacy rate, why can’t we? If we allow it, then we set up ourselves to have mediocre immune response.”
To note, Leachon was the Medical and Regulatory Affairs Director of Pfizer Philippines from 1995 to 2010.
Other ear-splitting politicians who have accused the Philippine government of prioritising Chinese developed vaccines, which is, of course, misleading and false include Bayan Muna party-list representative, Gabriela and party-list Rep. Arlene Brosas, both members of the “MAKABAYAN Bloc” in Congress, and Senator Riza Hontiveros who is a member of the “yellow” political opposition.
Explications And Elucidations
To put things into perspective and to clarify the misleading information and half-truths spread by the likes of Zubiri, Leachon, Gaite, Brosas, Hontiveros, and the PDI – a mainstream media outlet, what the DOST said is that China’s Sinovac Biotech COVID-19 vaccine may be deemed “acceptable” to minimum global standards, but the Philippines still needs to review the full data on it before it can be used.
The DOST also stressed that based on the WHO pronouncement, a 50 percent vaccine efficacy rate serves as the minimum requirement against the coronavirus. It contended that the scientific data of the Phase 3 trial of Sinovac has still to be reviewed and verified by the Vaccine Expert Panel before it could even be given an Emergency Use Authorization (EUA).
The DOST also further noted that the Philippines will not use or administer Sinovac or any COVID-19 vaccine until it gets final approval from the Food and Drug Administration (FDA).
Until such time that these processes have been completed, any conclusion as to the efficacy and efficiency of Chinese developed vaccines or any COVID-19 vaccine for that matter in the context of the Philippines and among Filipinos is not only premature but scientifically unwarranted and uncalled-for.
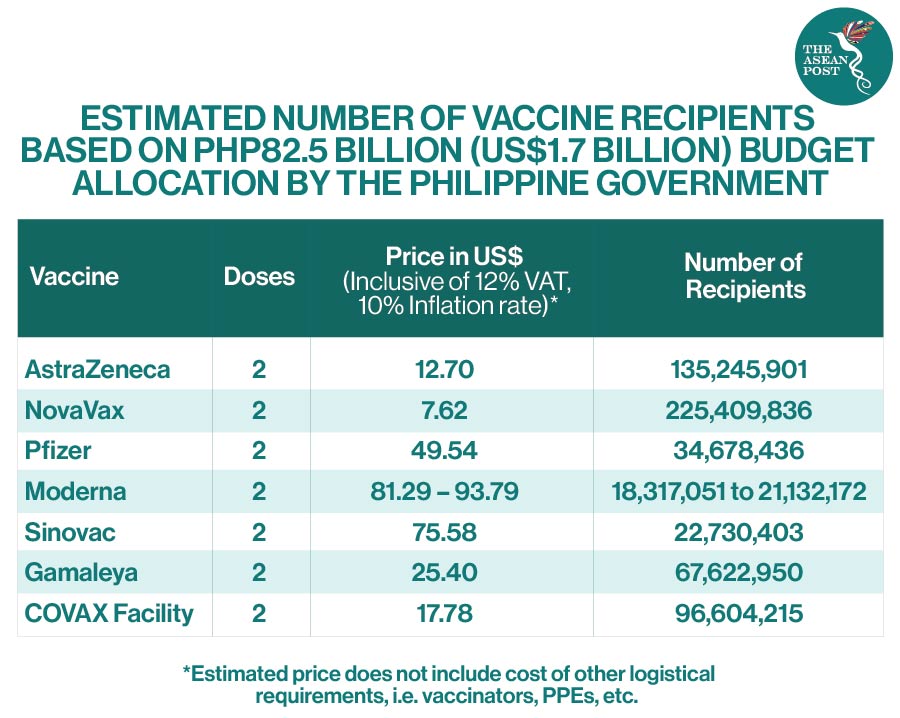
On another note, it is also not true that the Philippine government is only focusing on Chinese developed and manufactured COVID-19 vaccines. Vaccine Czar Carlito Galvez Jr. has several times explained to the public through various media interviews that the Philippine government at the moment is securing COVID-19 vaccine deals with America’s Johnson & Johnson and Moderna; British-Swedish firm AstraZeneca, Russia’s Gamaleya Institute, and India’s Novavax. US-based Pfizer has also applied for a EUA in the Philippines for its vaccine recently.
Galvez also reiterated several times in his media interviews that the country is conducting a “stringent” screening of vaccines, as prescribed by international institutions like the World Bank (WB), the Asian Development Bank (ADB), and Asian Investment Infrastructure Bank (AIIB), which are the primary sources of the country’s COVID-19 vaccine procurement financing.
Galvez said, “I would like to announce to our fellow Filipinos that the vaccine we choose is very safe and very effective considering that it will follow our stringent procedures and that of our fund manager or what we call procurement agents, which are ADB, WB, and the AIIB.”
Efficacy Vs Efficiency
Furthermore, it seems that the likes of senator Zubiri, Leachon, Gaite, Brosas, and Hontiveros are either confused or choosing to confuse the Filipino public by implying that the “efficacy and effectiveness” of a vaccine are one and the same. The two concepts are not the same, but rather refer to two entirely different things.
“Vaccine efficacy” refers to the degree to which a vaccine prevents disease, and possibly also transmission under ideal and controlled circumstances – comparing a vaccinated group with a placebo group, while “effectiveness” refers to how a vaccine performs in the real world.
According to the DOST, vaccine efficacy may change. It may go up or down as more people use it. Also, efficacy under laboratory conditions does not always translate into effectiveness once the vaccine has been rolled out in the real world. Thus, an efficacy trial can overestimate a vaccine’s impact in practice.
According to epidemiologists and vaccine experts, vaccines do not always need to have exceptionally high effectiveness to be useful. For example, the influenza vaccine is 40 – 60 percent effective, yet saves thousands of lives every year.
Sinovac Efficacy Rate In Question
On the issue of Sinovac’s efficacy rate, there’s a need to straighten out facts and information because of the misleading reporting by some mainstream media in the Philippines, which to a greater extent has contributed much to the disinformation, confusion, and misunderstanding among the Filipino public.
Researchers in Brazil have confirmed that the COVID-19 vaccine developed by China’s Sinovac Biotech Ltd showed efficacy between 50 percent and 90 percent in Brazilian trials. The full trial results will accordingly be released on 7 January, 2021. According to Brazil’s state-run Butantan Institute, the COVID-19 vaccine developed by China’s Sinovac achieved levels of efficacy against the coronavirus required by the WHO in trials with 13,000 Brazilian volunteers.
Dimas Covas, director of the Butantan Institute said that “the data corroborated that the vaccine is “the safest on the market” and has achieved the superiority in efficacy required by the WHO and Brazil’s Health Regulatory Agency (Anvisa).”
Covas added, “We have reached the level of effectiveness that allows us to request the registration for a EUA of the vaccine both here and in China. It is a historic day for Brazilian science because of the hope it brings to Brazilians.”
On another note, the Health Minister of Turkey, Fahrettin Koca also announced that the initial protection rate of the COVID-19 vaccine developed by Sinovac has a 91.25 percent efficacy rate and no major side-effects were seen during the trial. Koca further said that “We are sure that the vaccine (referring to Sinovac) is effective and safe for Turkish people.” He also announced that he will be among the first to be inoculated with the Chinese vaccine.
Thus far aside from Turkey and Brazil, Sinovac has secured supply deals for its vaccine with several countries including Indonesia, Chile, and Singapore, and is holding talks with the Philippines, while Malaysia is already in the final stage of negotiations with China-based manufacturers Sinovac and CanSino to secure doses of the vaccine to cover more of its population.
Conclusion
Without a doubt, the proliferation of false, half-truths, and misleading information and the politicisation of the COVID-19 vaccine procurement process has caused misunderstanding and fear among Filipinos.
This in many ways is not at all helpful, may cost lives, and has contributed much to public confusion, misunderstanding, and probably to the widespread aversion against vaccination. All these will not yield any benefits to the Philippines and its people in terms of ending the COVID-19 pandemic in the country.
One must understand that the value of any COVID-19 vaccine is very much dependent on the people’s willingness and enthusiasm to be vaccinated and that is very much dependent on how the public perceives the quality of the vaccine(s) to be procured.
Public perception is very much dependent and shaped by the information available. Thus, it is imperative that information consumed by the Filipino public is reliable, and reflective of the truth if the COVID-19 vaccination exercise is to be embraced by all Filipinos.
On this note, Filipinos must be discerning and exercise good judgment when assessing any information that’s in front of them and to always verify and double-check information before accepting them as the truth.
Filipinos should also be more careful with information they share on various social media platforms and to double-check first before sharing.
Furthermore, Filipinos should have more confidence and trust that the Philippine government is exerting all efforts to secure safe, effective, and cost-effective COVID-19 vaccines for everyone at the most appropriate time to eventually end the pandemic in the country.
Source: The ASEAN Post
https://theaseanpost.com/article/vaccine-politics-philippines



